 ILS Laboratories
ILS LaboratoriesVerified Certificate
Certificate of Analysis
All tests passed. This certificate is verified and authentic.
Tested For

LEO RESEARCH
www.leoresearchgroup.com
Product Information
Product
MT-2 - 10mg
Lot Number
050526
Appearance
Good
Test Type
Full QC Panel
Sample Received
05/05/2026
Analysis Confirmed
05/13/2026
Test Results — Full QC Panel
| Analyte | Limit | Result | Unit | Status |
|---|---|---|---|---|
| Purity (HPLC) | ≥ 95.0% | 99.84% | % | PASS |
| Net Peptide Content | Report Only | 10.41 | mg | N/A |
| Identity (ID) | MT-2 | Confirmed | - | PASS |
Elemental Impurities — ICP-MS per USP ⟨233⟩
| Element | Limit (ppm) | Result (ppm) | Status |
|---|---|---|---|
| Arsenic (As) | ≤ 1.5 ppm | 0.1108 ppm | PASS |
| Cadmium (Cd) | ≤ 0.5 ppm | 0.142 ppm | PASS |
| Chromium (Cr) | ≤ 10 ppm | 0.432 ppm | PASS |
| Mercury (Hg) | ≤ 1.5 ppm | 0.294 ppm | PASS |
| Lead (Pb) | ≤ 1 ppm | 0.1284 ppm | PASS |
Overall:PASS
Sterility Testing (PCR)
| Test | Specification | Result | Status |
|---|---|---|---|
| Sterility (PCR) | No Growth | No Growth | PASS |
Endotoxin Testing — USP ⟨85⟩
| Test | Specification | Result | Status |
|---|---|---|---|
| Endotoxin (USP <85>) | < 0.25 EU/mL | 0.09 EU/mL | PASS |
HPLC Chromatogram
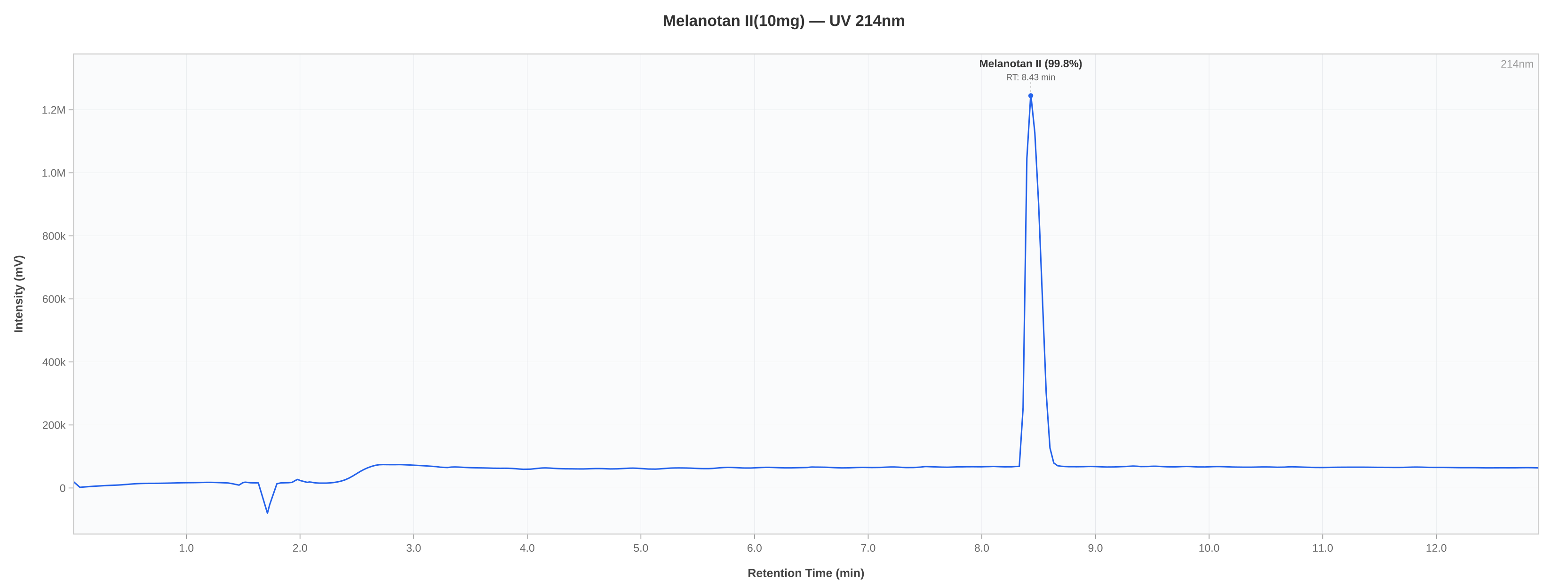
Notes & Methodology
- Sample confirmed to be MT-2 by HPLC. Identification by chromatographic retention time comparison with a reference standard.
- Elemental impurities analyzed by ICP-MS per USP ⟨233⟩.
- Bacterial endotoxin testing performed by Limulus Amebocyte Lysate (LAL) method per USP ⟨85⟩.
- Sterility testing performed by PCR (Polymerase Chain Reaction) method.
Dr. Greg Kalyuzhny
Lab Director
05/13/2026
COA: COA-2026-LQ30XN
Access Code: 14DDSDKO
Produced: 5/13/2026
This certificate was issued by ILS Laboratories LLC